Fiber and Colon Health On A Well-Formulated Ketogenic Diet: New Insights Question Its Role As An Unconditional Requirement

There are a few dietary recommendations that have been so ingrained in our minds that we accept them without question. The need for dietary fiber and the proposed benefits of a high fiber diet seem to be two of these, regardless of what type of diet we choose to eat. But why is fiber beneficial and how much do we need to get these claimed benefits? When we think of fiber in the diet we often think of vegetables, fruits, and whole grains, and that naturally begs the question, where does fiber fit into a well-formulated ketogenic diet? The answers to these questions are multi-faceted and potentially surprising.
Adults are generally encouraged to consume at least 25 to 30 grams of fiber per day. A well-formulated ketogenic diet (WFKD), while necessarily restricted in its carbohydrate content, can include several classes of foods that provide fiber (e.g., vegetables, seeds, nuts), but on average may only provide about half this amount. However, the nutritional ketosis resulting from a WFKD causes the liver to produce beta-hydroxybutyrate (BOHB), and BOHB has the potential to replace some of the functions of dietary fiber. As we will discuss, many of the benefits of fiber are attributable to its fermentation by bacteria which produce short-chain fatty acids (SCFAs) in the colon, especially one called butyrate. And, as it turns out, this SCFA has metabolic properties that are very similar to those of BOHB. Therefore, a well-formulated ketogenic diet may provide many of the benefits of fiber, without a high carbohydrate intake and the associated unpleasant side effects that some people experience from high fiber intake.
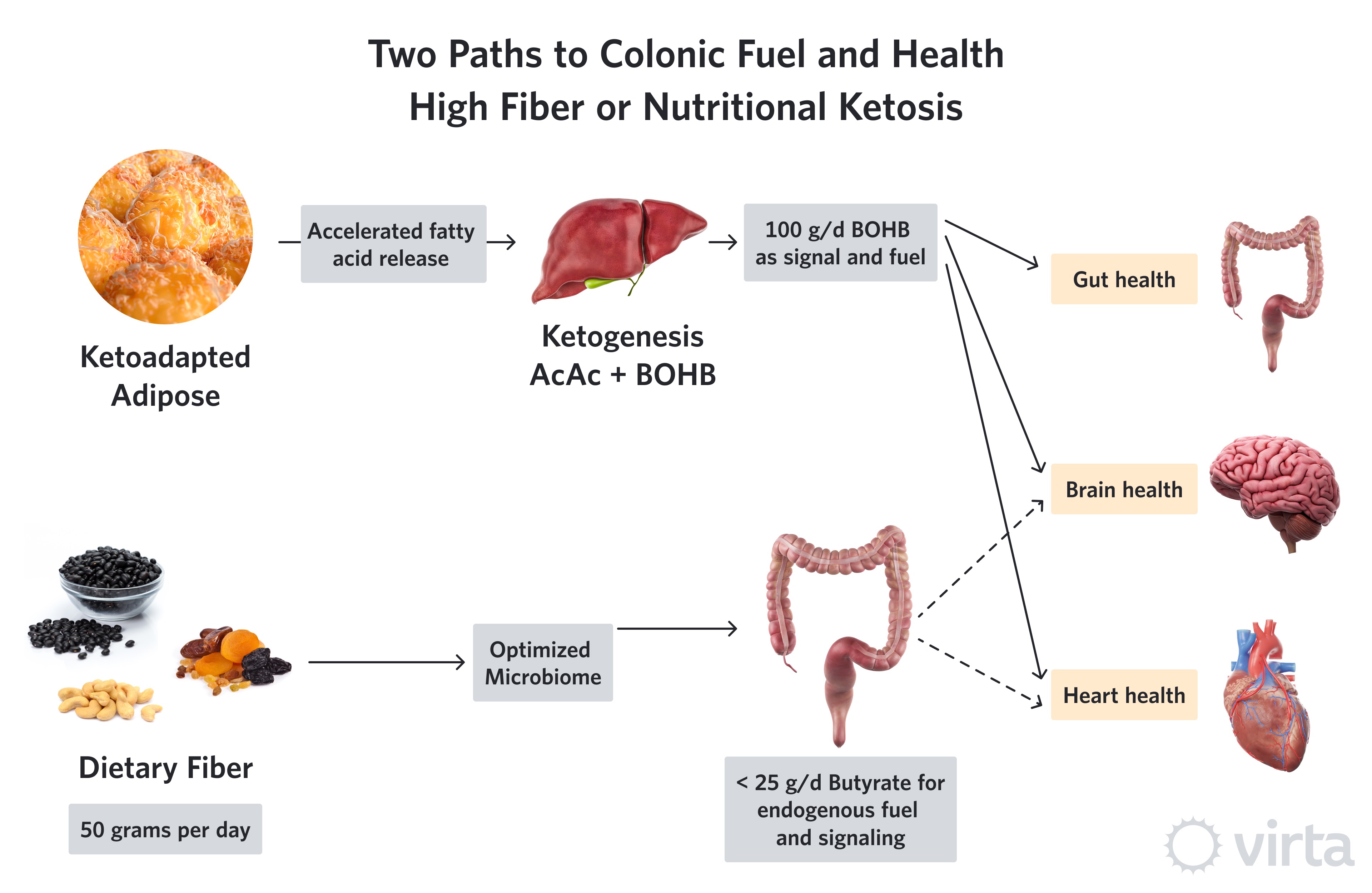
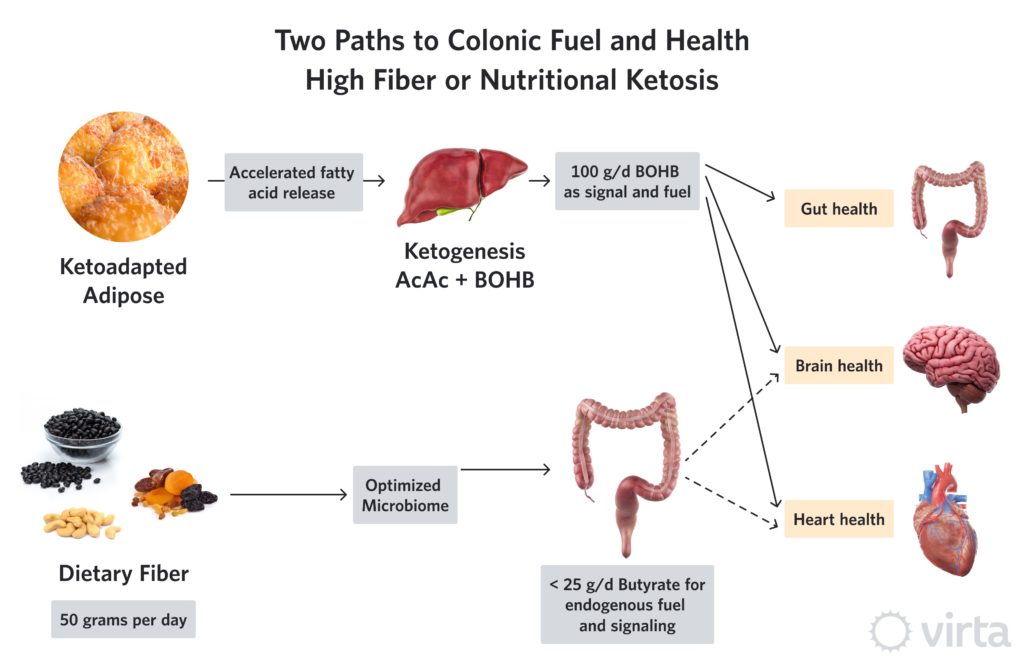
Snapshot: Two Pathways to Colonic Fuel and Health
As a result of consuming a ketone-suppressing diet (i.e., anything providing > 30% of daily energy need as carbohydrate plus protein), agricultural societies depend upon an optimized diet of fiber and colonic micro-organisms (aka, one’s own microbiome) to maintain colon health, and perhaps the health of other organs as well. This fact has led to a frenzy of research to find the optimal human microbiome—given the assumption that we all eat enough fiber as substrate to produce that microbiome’s desired product (butyrate). Butyrate is a preferred fuel (over glucose or longer fatty acids, as well as other SCFAs) to nourish the cells lining the large intestine (colon). But there is strong evidence that some of the butyrate produced by colonic fermentation is carried by the circulation throughout the body, thus improving the health of other organs like the heart and the brain¹.
Viewed from the perspective of the majority of the world’s population that now eats a ‘traditional’ Western diet, these systemic effects of colonic fiber fermentation are justifiably used as evidence supporting a high fiber diet, and in particular, a diet rich in long-chain polysaccharides known as soluble fiber. This substrate, in the context of an optimized microbiome, produces butyrate at a theoretical ratio of up to 49-to-100 (i.e., 49% by weight) from soluble fiber. Thus, a diet very high in soluble fiber providing 50 g/d could result in up to 24 g/d of colonic butyrate production. And given that only a fraction of that butyrate might escape into the systemic circulation (i.e, some is used by the colon and some extracted by the liver), the tangible benefits of a high fiber diet to other organs may be due to the fueling and signaling effects from no more than 10 g/d of butyrate.
Assuming that agriculture has been a dominant influence on human dietary choices for at most the last 10,000 years, whereas many human populations have been evolving away from the equatorial home of our ancestors for 2,500,000 years, this recent high carbohydrate phase of human evolution represents less than half of one percent of our metabolic past. And in fact, numerous nomadic hunting and herding cultures such as the Maasai, Mongols, and Great Plains Native Americans not only survived without dietary fiber—they thrived up to within the last 100-200 years. So how can some humans’ colons survive with little or no soluble dietary fiber?
We now know that a ketogenic diet can induce the liver to produce anywhere between 75 to 150 grams of beta-hydroxybutyrate (BOHB) every day. Chemically, BOHB is almost identical to butyrate with the exception of one added oxygen. Both butyrate and BOHB can be used by mitochondria as an efficient fuel, and both have anti-inflammatory and epigenetic effects. However, in this inflammation-modulating role, BOHB is more potent².
So, here’s our hypothesis: while the combination of high dietary fiber plus an optimum microbiome is critical to one’s health when eating a ketone-suppressing diet, this imperative decreases in importance in the context of a ketogenic diet. This is because adequate dietary carbohydrate restriction can induce the liver to produce many-fold more grams of BOHB than one could produce as butyrate via colonic fermentation. Thus, the keto-adapted individual whose liver is producing 100 g/d of BOHB—despite allocating around 50 g for the brain and 25 g for the heart—might still be able to allocate 25 g to maintain colon health and function without the need for appreciable colonic butyrate production from non-digestible carbohydrate fermentation.
Clearly there may be other benefits to the colon and systemic health from a high fiber diet. High-fiber ‘real foods’ (as opposed to most purified fiber supplements) are also rich in other phytonutrients and essential minerals. Our point here is not that dietary fiber is unimportant. To be specific, we are not challenging fiber's important role in colon health—along with an optimized microbiome—in the context of a diet whose combined total of carbohydrate and protein is high enough to suppress nutritional ketosis. However, cultural, historical, and now strong biochemical evidence indicates that many of the benefits of fiber fermentation in the colon can be replaced by the normal physiological production of BOHB by the liver during a well-formulated ketogenic diet.
Furthermore, since nutritional ketosis can be sustained by most people when consuming between 30 and 50 grams per day of total carbohydrate, that leaves plenty of room in one’s diet for fiber-rich foods such as non-starchy vegetables, nuts, seeds, and berry fruits. Consuming these foods has the added benefit of providing essential minerals such as potassium and magnesium. Thus, this is not a question of ‘either/or’ but one of crafting a well-formulated ketogenic diet (WFKD) that captures the benefits of liver ketone production plus a fair amount of dietary fiber—i.e., the best of both worlds.
What is Fiber?
Though the definition can be a bit nebulous, fiber is composed of various components of plants such as vegetables, grains, legumes, nuts, and fruits that are neither digested nor absorbed in the small intestine but are nonetheless able to be fermented by bacteria in the large intestine. Fiber is often further separated into two categories:
- Soluble (absorbs water, increases stool bulk, prone to fermentation)
- Insoluble (promotes motility, less prone to fermentation)
Most of us have heard of the benefits of fiber. After all, we have been told it is good for gut health, it may decrease our cholesterol and reduce our risk of type 2 diabetes, cardiovascular disease and some cancers as well as help us lose weight. This all sounds like good stuff! However, much of the considerable volume of published research on purified sources of fiber has failed to demonstrate clear benefits from the fiber itself, independent of other factors, in health outcomes³.
Because fiber is not officially considered an essential nutrient—that is, the body can function without dietary intake of fiber—there is no official recommended daily allowance (RDA) for fiber in the United States. There are some general guidelines however, that suggest an average Daily Value (DV) of about 25g or more per day.
Despite the emphasis placed on fiber as a part of a healthy diet (assuming that one is eating lots of dietary carbohydrate and thus not in nutritional ketosis), the fact remains that fiber is not an essential nutrient in its own right. From this perspective, soluble fiber plays a supporting role in colon health, but only if one has an optimized microbiome that produces butyrate. Thus, while it tends to get top billing in health promotion, since fiber primarily facilitates the production of SCFAs like butyrate, they are the real stars of the show.
Short-Chain Fatty Acids
Three SCFAs—acetate, propionate and butyrate—are produced in the intestinal lumen by bacterial fermentation of dietary fiber. SCFAs serve as energy substrates for the gut and perform other beneficial actions for gut and overall health. Butyrate is the most extensively-researched of the SCFAs and is implicated in many of the health-promoting effects associated with colonic fermentation of fiber. The SCFA butyrate also has some pretty potent anti-inflammatory properties that enhance the intestinal barrier, mucosal immune function⁴ and cellular energy metabolism⁵.
One of these powerful functions of butyrate is its action as a histone deacetylase (HDAC) inhibitor⁶. HDACs are enzymes that regulate specific genes (i.e., epigenetic regulation) of target cells, and inhibition of them by butyrate can help reduce unwanted cell growth and oxidative stress. And, for those of you familiar with our prior post about BOHB, you will recall that BOHB is also an HDAC inhibitor⁷. Both butyrate and BOHB bind to the same cell surface receptor in the colon, which means they can have similar tumor suppressing effects upon binding. This is likely one of the underlying mechanisms by which fiber intake is understood to contribute to a reduction in the incidence of colon cancer.
The Influence of Fiber on Components of Health
We mentioned some of the benefits that have been attributed to fiber including: improved gut health and motility, reduced risk of CVD, improved glycemia and insulin sensitivity, as well as help with weight control. How fiber contributes to these aspects is likely multifactorial, but one common thread throughout all of them is the action of butyrate. Interestingly, similar, and in some cases more robust, improvements in these same outcomes are seen in individuals who are in nutritional ketosis.
Gut Health and Motility. Gut health and the involvement of the gut microbiome in overall health, immune function and the modulation of disease are topics of increasing research interest. Colonic fermentation of fiber can influence gut motility, provide substrate for the cells of the gut lining and exert anti-cancer effects, in large part due to butyrate and its role in HDAC inhibition⁸. And, in certain circumstances, butyrate may work synergistically with the gut microbiome to exert these effects.
While fiber is thought to increase gut motility and ease of bowel movements, (again, butyrate is to thank for this as it regulates fluid and mineral balance) the effect of minerals like magnesium and sodium, as well as hydration status, cannot be overstated. Stool water content significantly effects motility. This is especially true for those on a WFKD. Careful attention to magnesium, sodium and fluid intakes can improve constipation, whereas in some instances, fibrous foods can actually worsen constipation symptoms⁹.
Cardiovascular Health. Fiber has been described as a potential aid in reducing cardiovascular disease based largely on epidemiological associations that have been reported between insoluble whole-grain cereal fibers and a reduction in coronary heart disease (CHD) risk¹⁰. However, despite this pervasive recommendation to increase fiber intake for heart health, the strength of these and other epidemiological relationships used to shape dietary advice is increasingly being called into question¹¹. When compared to results from controlled studies looking exclusively at fiber, the results often fail to show the same relationships. The challenge, one that is especially relevant to fiber containing foods, is teasing out the effects of fiber alone—independent of other constituents of the whole food—on health measures. The influence of confounding variables, or other factors that may significantly alter the reported outcomes, are often not taken in to account when reporting the results of observational research.
When we look more closely at the effect of fiber on CHD, we need to consider factors such as increased SCFA production and decreased caloric intake, as well as increased antioxidants, vitamins and minerals¹². These factors are known to influence inflammation, which is increasingly acknowledged to be a driver in the development of CHD¹³. In fact, a sustainable diet that reduces inflammation¹⁴ may prove to be more beneficial for cardiovascular health than a reductionist focus on fiber per se.
In terms of cholesterol, while dietary fiber is purported to lower cholesterol via binding of bile acids in the intestine leading to decreased absorption and increased elimination, careful research has failed to consistently show that this translates to significant long-term changes in serum cholesterol levels ¹⁵,¹⁶.
Type 2 Diabetes and Weight Control. The increasing incidence of metabolic disease worldwide has sharpened the focus on avenues to mitigate this growing health crisis. Because diet and lifestyle interventions are a first line of defense, identifying dietary components that promote metabolic health have become a considerable priority. Fiber is frequently described as one way to use the diet to positively affect glycemia and weight control. The challenge again becomes identifying the specific aspect of fiber-rich foods, and/or the specific types of fiber(s) that might be capable of these effects, because not all fiber acts the same way.
Additionally, there is a paucity of evidence to show that the lower glycemic response observed with acute fiber consumption continues over time in a manner that contributes to long-term weight control in humans¹⁰. Further, most well-designed studies have not shown significant, sustained weight reduction that is attributable to fiber in individuals consuming high fiber real-foods³. The inclusion of plant-based foods may provide other health promoting effects, but fiber itself has not been shown to be the causal factor in weight control.
Research into the impact of dietary fiber specifically on parameters related to the treatment and/or prevention of type 2 diabetes (T2D) have produced mixed results¹⁷. Interestingly, individuals with T2D have fewer butyrate-producing bacteria in their gut, suggesting a potential protective role of butyrate in this disease¹⁸. Because insulin resistance plays a significant role in T2D, factors that improve insulin resistance can have positive implications for patients with T2D. While some prospective cohort studies report associations between improvements in insulin resistance and increased intake of insoluble fibers, the same relationships have not been seen for soluble fibers from fruits and vegetables¹⁰. These differences may be due, in part, to the digestive properties of these different types of fiber, as well as the obligate role of an optimized microbiome in addition to the fiber. However, based on current evidence, it is difficult to attribute a significant impact in improving T2D to fiber alone.
Gut Microbiome. New research is continuing to emerge regarding the effects of a ketogenic diet on the gut microbiome. It has been shown that diet can dramatically alter the content of the gut microbiota¹⁹ and the microbiome can affect everything from the actions of medications²⁰ and the immune response²¹ to neural functioning ²²,²³, mental health²⁴, and bone homeostasis²⁵. The microbiota’s influence is multifactorial and may adjust to the composition of the diet in response to both ingested microbes and to compensate for nutrient needs¹⁹, allowing it to work synergistically to effect health outcomes.
The gut microbiota is also a potential factor in obesity and T2D as well as auto-immune diseases and other conditions. Therefore, as we continue to learn more about the role the microbiome plays in the modulation of health and disease, we will learn how the composition of the diet can influence these outcomes.
Dietary Fiber in the Context of a Ketogenic Diet. As we look at the proposed benefits of fiber, we can see that for those individuals who are on a WFKD, many of these positive effects are also achieved, or enhanced, as a result of being in nutritional ketosis. We want to emphasize that this is not to say that fiber is unnecessary as much as it is to say that butyrate appears to be the powerful actor behind fiber. Thus, in the context of a WFKD, the internal production of BOHB by the liver can reduce or eliminate the need for butyrate to achieve the desired health outcomes.
As a result, the need for a high dietary fiber intake may in fact be conditional and based upon the overall and consistent composition of the diet. If an individual is in nutritional ketosis, it may not be necessary to consume a high fiber diet (>25g per day) to achieve the health effects attributed to fiber. That said, while the fiber itself may be conditionally necessary, the additional nutrients and minerals provided by many fiber-rich foods may provide additional benefits and can certainly be a part of a WFKD.
Unfortunately, there is not much published research that has focused on determining the optimal amount of fiber to consume in the context of a WFKD. Dr. Volek and a doctoral student at the time (Dr. Richard Wood) published a series of studies aimed at determining the impact of adding a daily soluble fiber supplement in the form of glucomannan (a highly viscous soluble fiber) to a ketogenic diet²⁶,²⁷,²⁸. The ketogenic diet resulted in significant improvements in weight loss, body composition, blood pressure, glucose, cholesterol and lipoprotein profiles, and inflammatory markers; however, there was no additional benefit of increasing soluble fiber beyond the diet alone.
Wrap up: Why Standard Fiber Recommendations Do Not Apply to a WFKD
We are repeatedly presented with “new” epidemiological research regarding the role of various aspects of our diet and their influence on specific disease risks and long-term health. In spite of the often bold claims, there is increasing evidence that caution is required when drawing broad health recommendations based on associations derived from observational studies¹¹,²⁹. While this type of research can provide value from both the duration of an observed association and in identifying potential areas needing prospective research confirmation, it should not be the sole basis used to conclude causation. Thus, a realistic scientific perspective of dietary recommendations drawn from epidemiological associations might be summarized as “Trust Conditionally, But Then Verify.”
This wide-reaching concern with conclusions drawn from epidemiology is particularly relevant to questions about the health effects of ketogenic diets. Nutritional ketosis is a unique metabolic state that is increasingly recognized as having potent beneficial effects on energy metabolism, metabolic health, inflammation, gut health, and even positively influencing the gut microbiome. Given that virtually every large population study that has assessed associations between dietary choice and disease risk has not included a significant number of people following a well-formulated ketogenic diet, conclusions about the health effects of a ketogenic diet from such studies have no scientific basis. Specifically, associations linking poor outcomes to limited dietary fiber or somewhat increased fat consumption that are clearly well outside the range that is characteristic for sustained nutritional ketosis need to be viewed with considerable caution. That said, the fact that most individuals can nonetheless sustain nutritional ketosis while simultaneously consuming fiber from real foods at or above 25 grams per day (Table 1) means they can benefit from the best of both worlds.
Table 1

This blog is intended for informational purposes only and is not meant to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or any advice relating to your health. View full disclaimer
Are you living with type 2 diabetes, prediabetes, or unwanted weight?

- Crawford, PA.,Crowley, JR.,Sambandam, N. et al. Regulation of myocardial ketone body metabolism by the gut microbiota during nutrient deprivation. PNAS. 2009;106(27):11276-11281
- Youm, Y-H., Nguyen, KY., Grant, RW., et al. Ketone body β-hydroxybutyrate blocks the NLRP3 inflammasome-mediated inflammatory disease. Nat Med. 2015; 21(3): 263–269
- Brownlee, IA., Chater, PI., Pearson, JP., et al. Dietary fibre and weight loss: Where are we now? Food Hydrocolloids. 2017; 68:186-191
- Sivaprakasam, S.,Bhutia, YD.,Ramachandran,S., et al. Cell-Surface and Nuclear Receptors in the Colon as Targets for Bacterial Metabolites and Its Relevance to Colon Health. Nutrients. 2017;9 (856):1-15
- Kootte, RS., Vrieze, A.,Holleman, F. et al. The therapeutic potential of manipulating gut microbiota in obesity and type 2 diabetes mellitus. Diabetes, Obesity and Metabolism. 2012; 14:112–120
- Chang, PV.,Hao, L.,Offermanns. S., et al. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. PNAS. 2014;111(6):2247-2252
- Newman, J., Verdin, E. β-hydroxybutyrate: Much more than a metabolite. Diabetes Res Clin Pract. 2014;106(2): 173–181.
- Flint, H.J., Scott, K.P., Louis, P., and Duncan, S.H. (2012). The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 9, 577–589.
- Kok-Sun Ho, Charmaine You Mei Tan, Muhd Ashik Mohd Daud, et al. Stopping or reducing dietary fiber intake reduces constipation and its associated symptoms. World J Gastroenterol. 2012; 18(33): 4593-4596
- Weickert, MO., Pfeiffer, AFH. Impact of Dietary Fiber Consumption
on Insulin Resistance and the Prevention of Type 2 Diabetes. J Nutr 2018;148:7–12 - Ioannidis, JPA. The Challenge of Reforming Nutritional Epidemiologic Research. JAMA. 2018;320(10):969-970
- Lattimer, JM., Haub, MD. Effects of Dietary Fiber and Its Components on Metabolic Health. Nutrients. 2010; 2:1266-1289
- Libby, P. Inflammation and cardiovascular disease mechanisms. Am J Clin Nutr 2006;83(suppl):456S–60S
- Bhanpuri, NH.,Hallberg, SJ.,Williams, PT., et al. Cardiovascular disease risk factor responses to a type 2 diabetes care model including nutritional ketosis induced by sustained carbohydrate restriction at 1 year: an open label, non-randomized, controlled study. Cardiovasc Diabetol.
- Van Rosendaal, GMA., Shaffer, EA., Edwards, AL., et al. Effect of time of administration on cholesterol-lowering by psyllium: a randomized cross-over study in normocholesterolemic or slightly hypercholesterolemic subjects. Nutrition Journal. 2004;3:17
- Jenkins, DJA., Kendall, CWC., Axelsen, M.,et al. Viscous and nonviscous fibres, nonabsorbable and low glycaemic index carbohydrates, blood lipids and coronary heart disease. Current Opinion in Lipidology. 2000;11:49±56
- Thompson SV, Hannon BA, An RP, et al. Effects of isolated soluble fiber supplementation on body weight, glycemia, and insulinemia in adults with overweight and obesity: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. 2017; 106:1514–1528
- Qin,J., Li, Y.,Cai, Z., et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490:55-60
- David, LA., Maurice, CF., Carmody, RN. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014; 505(7484): 559–563
- Clayton, TA., Baker, D., Lindon, JC., et al. Pharmacometabonomic identification of a significant host-microbiome metabolic interaction affecting human drug metabolism. PNAS. 2009; 106(34): 14728–14733
- Thaiss, CA., Zmora, N., Levy, M., et al. The microbiome and innate immunity. Nature. 2016; 535:65-74
- Olson, CA., Vuong, HE.,Yano, JM., et al. The Gut Microbiota Mediates the Anti-Seizure Effects of the Ketogenic Diet. Cell. 2018;173:1728–1741
- Bourassa, MW.,Alimab, I.,Bultman, SJ., et al. Butyrate, neuroepigenetics and the gut microbiome: Can a high fiber diet improve brain health? Neuroscience Letters. 2016; 625, 20:56-63
- Rieder, R., Wisniewski, PJ., Alderman, BL., et al. Microbes and mental health: A review. Brain, Behavior, and Immunity. 2017; 66:9-17
- Lucas, S., Omata, Y., Hofmann, J., et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nature Communications. 2018; 9(55):1-10
- Wood RJ, Fernandez, ML., Sharman, MJ., et al. Effects of a carbohydrate-restricted diet with and without supplemental soluble fiber on plasma low-density lipoprotein cholesterol and other clinical markers of cardiovascular risk. Metabolism. 2007;56(1):58-67
- Wood RJ, Volek JS, Liu Y, et al. Carbohydrate restriction alters lipoprotein metabolism by modifying VLDL, LDL, and HDL subfraction distribution and size in overweight men. J Nutr. 2006;136(2):384-9
- Wood RJ, Volek JS, Davis SR, et al. Effects of a carbohydrate-restricted diet on emerging plasma markers for cardiovascular disease. Nutr Metab (Lond). 2006;3:19
- Ioannidis, JPA. Why Most Published Research Findings Are False. PLoS.2005; 2(8):e24









